Most-download articles are from the articles published in 2024 during the last three month.
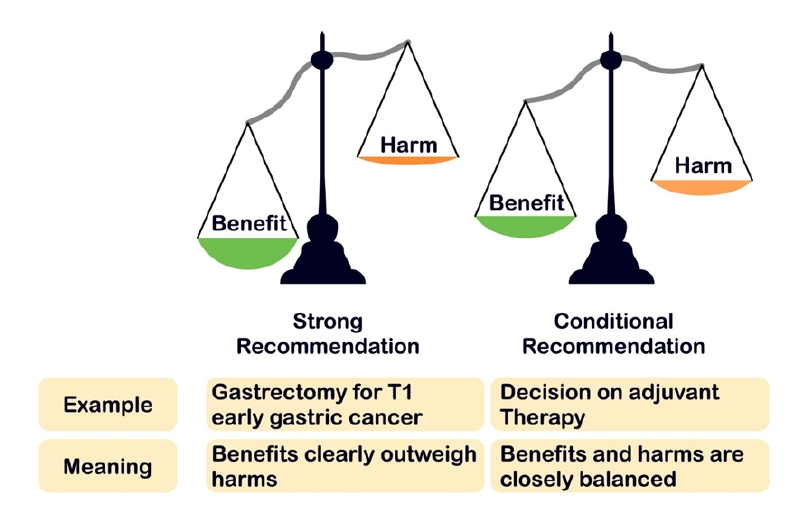
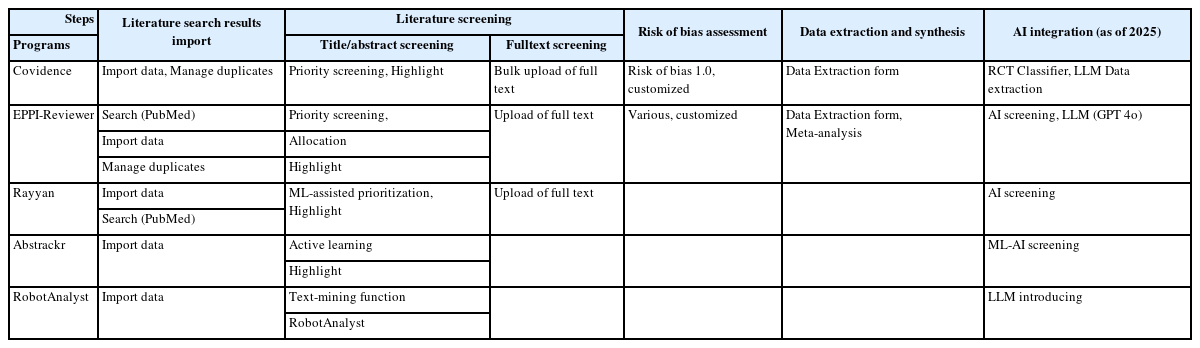
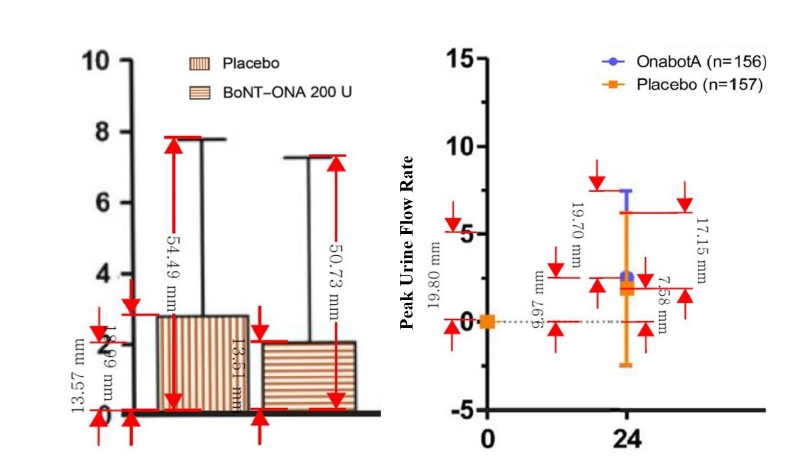
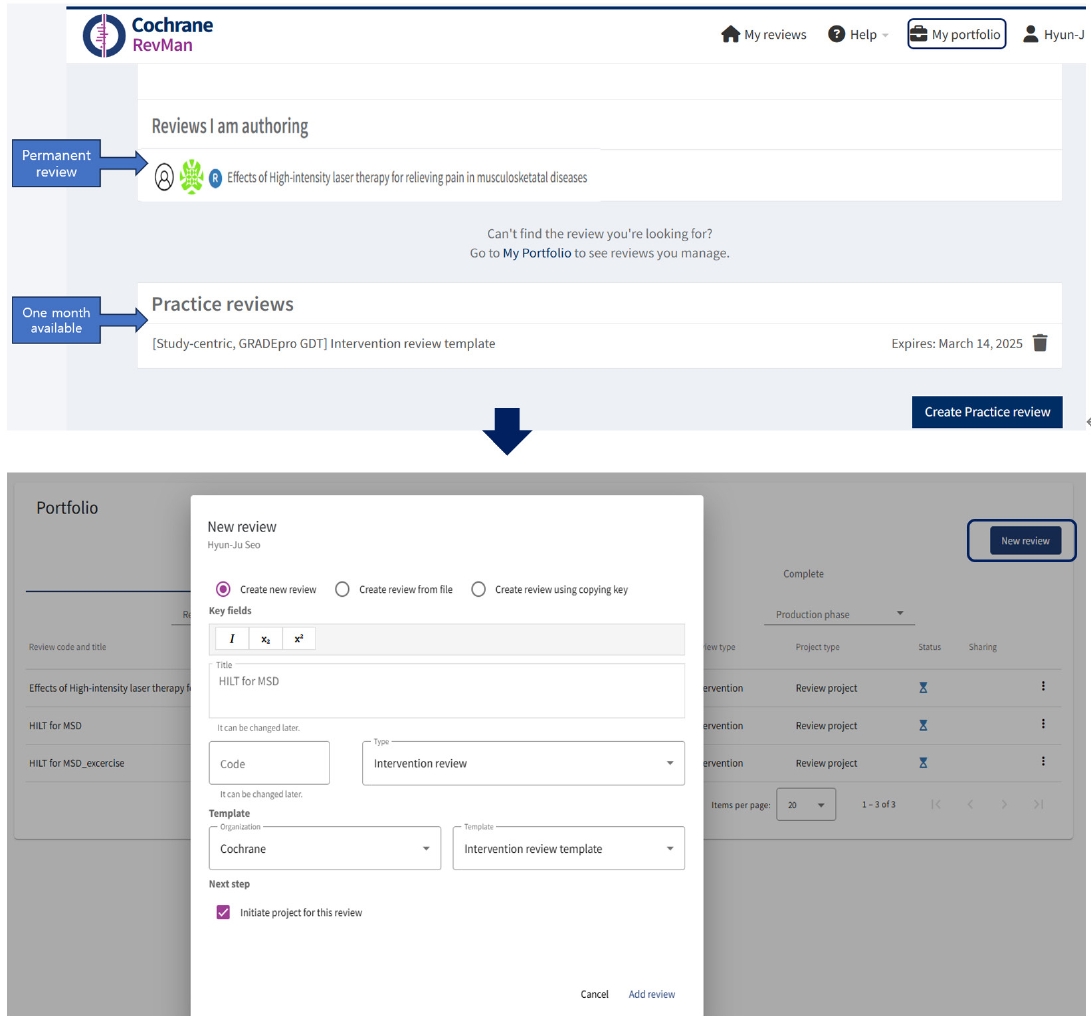
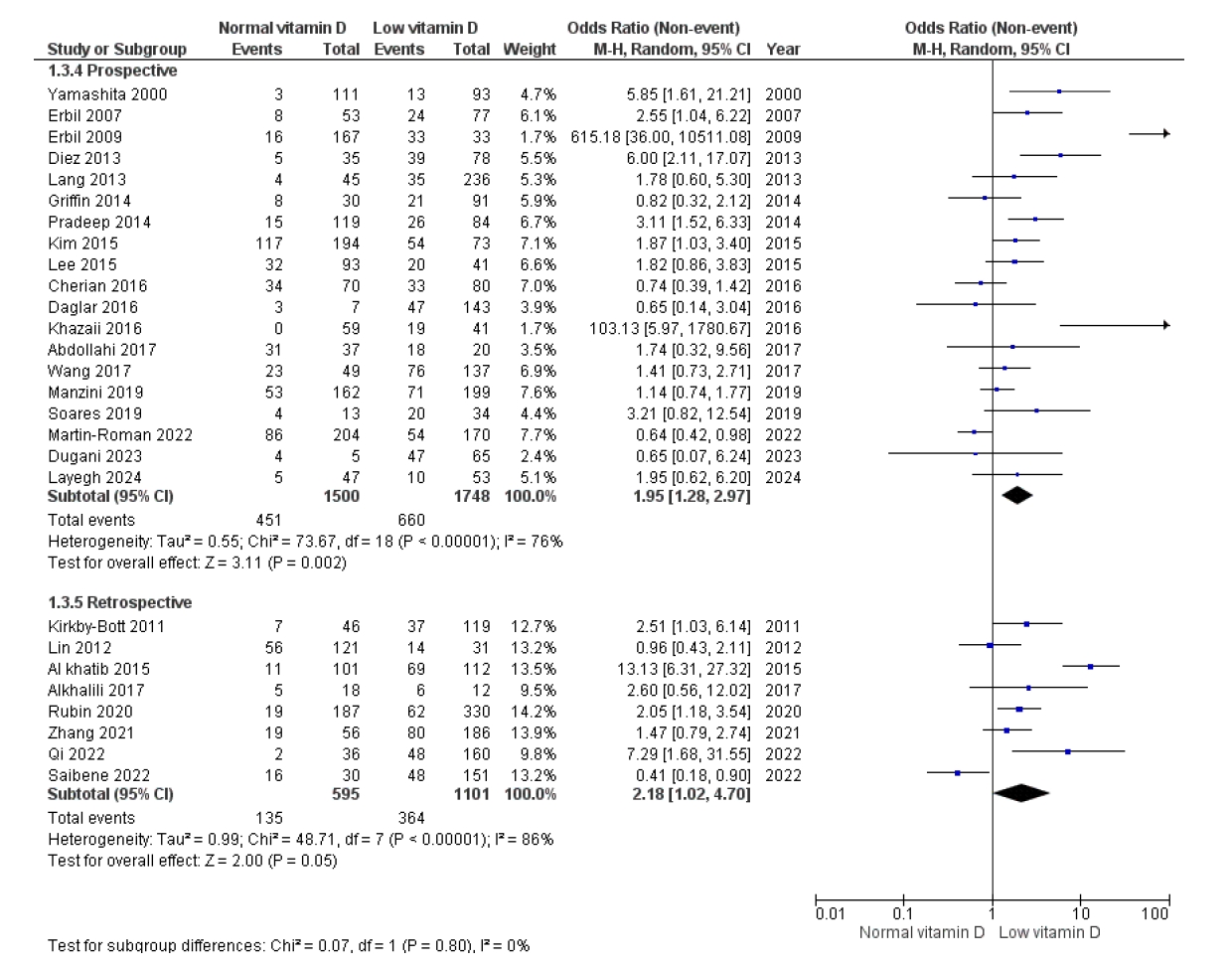
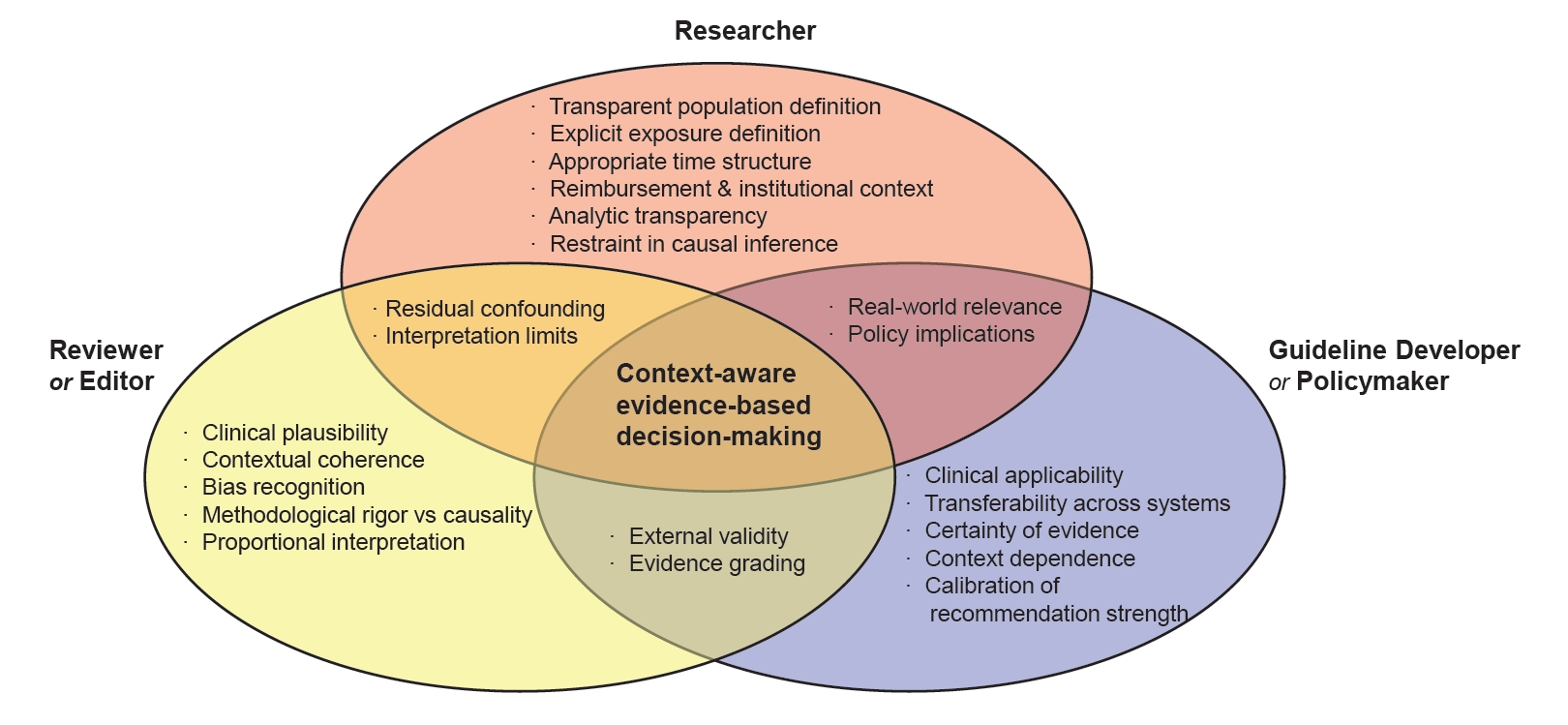
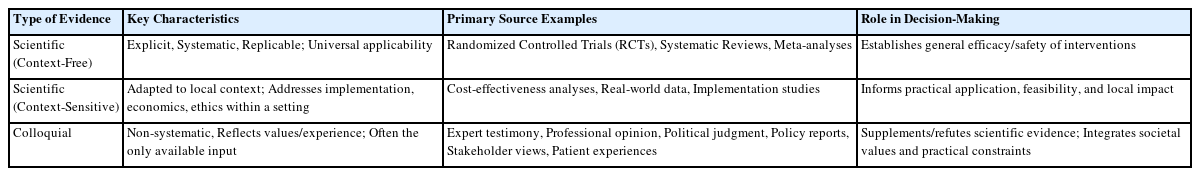
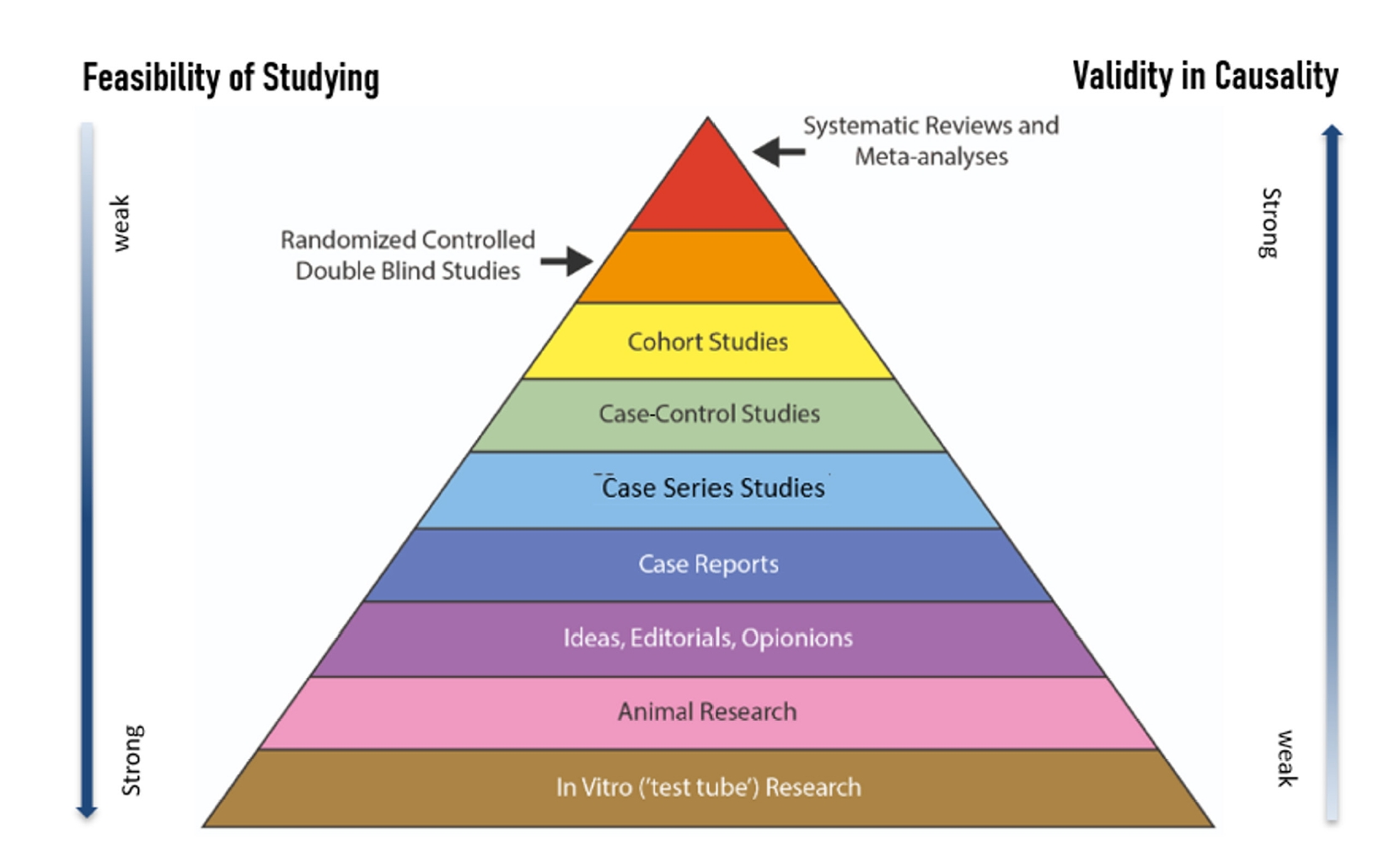
Preprints have become a transformative tool in scientific communication, addressing critical challenges of traditional publishing, including long peer-review timelines, high costs, and systemic publication bias. Publication bias, which disproportionately favors studies with positive or statistically significant results, undermines the comprehensiveness and accuracy of the scientific record. By offering an open platform for sharing all research findings, preprints ensure that studies with null or negative results are also represented, mitigating the selective publication that skews research fields and meta-analyses. The COVID-19 pandemic highlighted the importance of preprints, as they facilitated the rapid dissemination of urgent findings while maintaining accessibility. Unlike traditional journals, preprints bypass lengthy review processes, enabling immediate access to data and fostering timely feedback, collaboration, and application. This inclusivity and transparency enhance trust in the research process while democratizing access to scientific knowledge. Despite their advantages, preprints face challenges, such as inconsistent quality standards, discrepancies between preprints and final publications, and risks associated with unverified findings. These challenges can complicate their use in systematic reviews and evidence-based medicine, requiring careful consideration and handling.This paper explores the interplay between preprints and publication bias, detailing how preprints can reduce bias while identifying limitations that must be addressed.


